 For archaeological periods and date ranges for the Jordan Valley, see Supporting Information, Text S1. This investigation focuses on the Middle Bronze Age II (MB II) from ~ 1800–1550 BCE. More than just a mere city, TeH comprised the urban core of a city-state that flourished nonstop for ~ 3000 years during the Chalcolithic Period and Bronze Age beginning ~ 4700 BCE until it was destroyed at ~ 1650 BCE (3600 cal BP). The site contains the stratified remains of a fortified urban center, now known as the largest continuously occupied Bronze Age city in the southern Levant 1. A mound of ancient ruins is referred to as “tel” in Hebrew and “tell” or “tall” in Arabic. TeH is a raised, two-tiered occupational mound, the largest in the Jordan Valley. Tunguska-scale airbursts can devastate entire cities/regions and thus, pose a severe modern-day hazard. Tall el-Hammam may be the second oldest city/town destroyed by a cosmic airburst/impact, after Abu Hureyra, Syria, and possibly the earliest site with an oral tradition that was written down (Genesis). 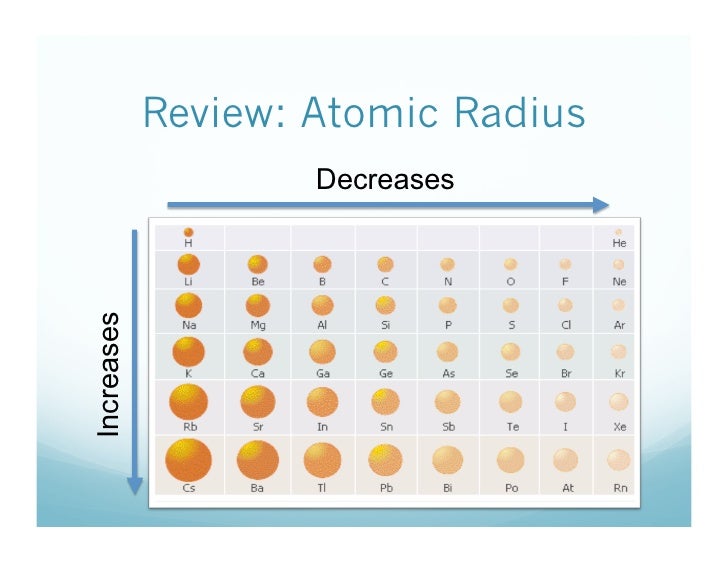
An airburst-related influx of salt (~ 4 wt.%) produced hypersalinity, inhibited agriculture, and caused a ~ 300–600-year-long abandonment of ~ 120 regional settlements within a > 25-km radius. Amid city-side devastation, the airburst demolished 12+ m of the 4-to-5-story palace complex and the massive 4-m-thick mudbrick rampart, while causing extreme disarticulation and skeletal fragmentation in nearby humans. Heating experiments indicate temperatures exceeded 2000 ☌. A city-wide ~ 1.5-m-thick carbon-and-ash-rich destruction layer contains peak concentrations of shocked quartz (~ 5–10 GPa) melted pottery and mudbricks diamond-like carbon soot Fe- and Si-rich spherules CaCO 3 spherules from melted plaster and melted platinum, iridium, nickel, gold, silver, zircon, chromite, and quartz. The proposed airburst was larger than the 1908 explosion over Tunguska, Russia, where a ~ 50-m-wide bolide detonated with ~ 1000× more energy than the Hiroshima atomic bomb. It does not store any personal data.We present evidence that in ~ 1650 BCE (~ 3600 years ago), a cosmic airburst destroyed Tall el-Hammam, a Middle-Bronze-Age city in the southern Jordan Valley northeast of the Dead Sea. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Terms in this set (6) Of the elements calcium, Ca, beryllium, Be, barium, Ba and strontium, Sr, which has the largest atomic radius? Barium has the highest atomic number, so barium has the largest atomic radius because atomic radii increase down a group. Which has a larger radius beryllium or barium? (A) Comparison of Atomic and Ionic Radius of Group 1 (IA, alkali metals) Elements ElementĬonsequently, the ion with the greatest nuclear charge (Al3+) is the smallest, and the ion with the smallest nuclear charge (N3−) is the largest….Ionic Radii and Isoelectronic Series. Which element has the largest radius Na or CS? These are (except for hydrogen) soft, shiny, low-melting, highly reactive metals, which tarnish when exposed to air. Group 1A (or IA) of the periodic table are the alkali metals: hydrogen (H), lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). Which element is an alkali metal Rb or Al?  
Atomic size decreases when going from left to right across a period Atomic size increases when going from top to bottom down a group. 
Which of the following atoms has the largest atomic radius a GE B NA c rb d/s e br? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
